
Clinical trials are essentially the means through which new medical innovations reach the market. The outcome of a clinical trial, be it a success or a failure, is largely dependent on a single factor: site selection.
Apart from that, the correct clinical trial sites basically serve as the engine that not only powers patient enrollment rates but also helps to maintain data quality, compliance, and the overall study timeline.
These days, there is a lot of competition for sites among researchers, and at the same time, regulatory requirements are becoming stricter, so it is vital to know the clinical trial site selection process.
Site selection in clinical trials entails a strategic endeavor that involves, among other things, pinpointing potential locations for clinical research, scrutinizing, and finally deciding on those sites that are well-equipped with proper infrastructure and have access to a suitable patient population; furthermore, the staff should have the required expertise, and the site should be operationally capable of carrying out the study successfully.
An effective site selection process in clinical trials leads to the smooth, ethical, and globally regulated execution of the study.
Selecting appropriate clinical trial sites is a must for a variety of reasons:
The chosen site is the one that lays the groundwork for the whole study. A bad site decision will impact everything, from timelines to data integrity.
The correct site leads to:
Basically, choosing a site is the most strategically pivotal decision that sponsors make.
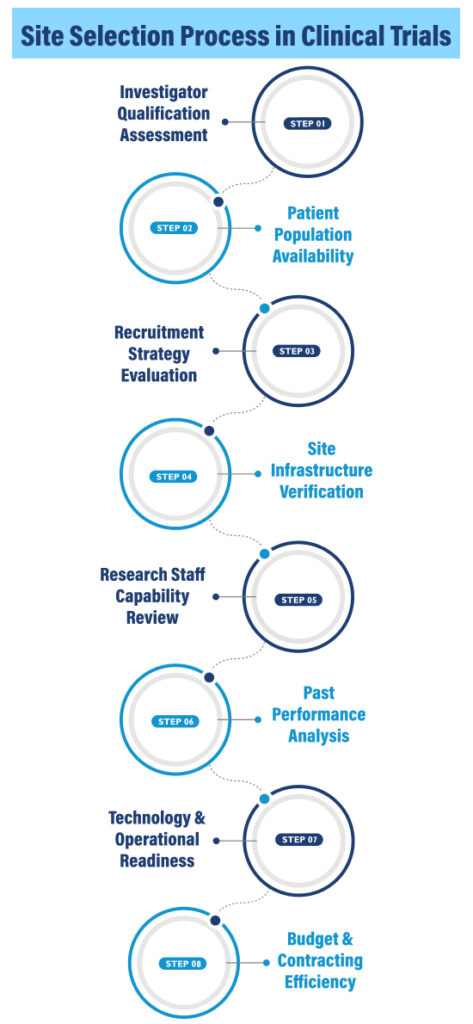
Selecting the right clinical trial site requires a structured evaluation of expertise, infrastructure, patient access, and operational readiness. A systematic process helps sponsors identify sites that can deliver quality data, timely recruitment, and regulatory compliance.
Step 1: Investigator Qualification Assessment
Step 2: Patient Population Availability
Step 3: Recruitment Strategy Evaluation
Step 4: Site Infrastructure Verification
Step 5: Research Staff Capability Review
Step 6: Past Performance Analysis
Step 7: Technology & Operational Readiness
Step 8: Budget & Contracting Efficiency
Those sites that deliver top performance are usually fully booked and may not have the capacity to accommodate new studies.
The initiation of a research project is brought to a standstill by the delay in the contract, budget, and ethics approval stages.
Failure to enrol enough participants on time is the major cause for clinical trials missing deadlines.
Locations sometimes portray that they have the ability to do more than they actually can or that patient enrollment potential is higher than it really is.
The site may have lapses in the documentation, auditing, or training areas.
Clinvigilant Research supports sponsors to make well-informed decisions by providing:
1. Global Feasibility Assessment
Use of worldwide site networks, investigator databases, and historical performance metrics.
2. End-to-End Site Evaluation Support
Clinvigilant Research moves with the utmost efficiency from feasibility to SSVs, therefore, ensuring that only the most qualified sites are chosen.
3. Faster Start-Up Timelines
The initiation of the site is made very fast through the implementation of streamlined contracts, regulatory submissions, and ethics coordination
4. Technology-Driven Site Management
Operations that are remote monitoring, digital documentation, EDC support, and rapid communication cycles are the technologies that allow the smooth execution of activities.
5. Better Enrollment and Data Quality
Clinvigilant Research speeds up participant recruitment by selecting high-performing sites. Moreover, it makes sure that the data is clean and reliable.
Site selection means the process of determining and assessing research sites that are able to carry out a clinical study safely and efficiently.
Site selection is important because it depends on patient recruitment, data quality, compliance, and overall study timelines.
Character of the Investigator – relevant experience, availability of patients, appropriate infrastructure, recruitment potential, and good performance history.
A feasibility questionnaire is a detailed assessment tool used by a sponsor to evaluate a clinical study site’s capabilities, experience, and readiness.
Clinvigilant helps to ensure successful study execution by supporting feasibility, global site identification, selection, start-up, and ongoing oversight.
It is a visit to a research site by a CRA to check if the site has the facilities it claims, correct staffing, proper documentation practices, and if the site is responding genuinely to a feasibility questionnaire.
Site selection ranks among the most pivotal processes through the entire clinical trial lifecycle. An effectively chosen research site has the power to not only shorten the timelines but also to improve the patient enrollment, enhance the data accuracy, and elevate the chances of study success.
In the face of a continually changing clinical research environment, sponsors are required to take a strategic and data-driven approach to the site selection process in clinical trials.
Collaborating with an expert CRO like Clinvigilant Research ensures access to high-performing sites, rigorous feasibility assessments, and streamlined study start-up, all of which drive superior clinical outcomes.

Clinvigilant Research is a global full-service CRO (Contract Research Organization) dedicated to advancing clinical development with scientific precision and patient-centric solutions. With our end-to-end CRO services sponsors can accelerate time-to-market while maintaining the highest standards of quality and compliance. With global reach and deep therapeutic expertise, we partners with sponsors to bring safe, effective, and innovative therapies to patients worldwide.
| Cookie | Duration | Description |
|---|---|---|
| _wpfuuid | 11 years | This cookie is used by the WPForms WordPress plugin. The cookie is used to allows the paid version of the plugin to connect entries by the same user and is used for some additional features like the Form Abandonment addon. |
| cookielawinfo-checkbox-advertisement | 1 year | Set by the GDPR Cookie Consent plugin, this cookie is used to record the user consent for the cookies in the "Advertisement" category . |
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| CookieLawInfoConsent | 1 year | Records the default button state of the corresponding category & the status of CCPA. It works only in coordination with the primary cookie. |
| elementor | never | This cookie is used by the website's WordPress theme. It allows the website owner to implement or change the website's content in real-time. |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |
| Cookie | Duration | Description |
|---|---|---|
| GetLocalTimeZone | session | No description |